Product
Efficiency in Action
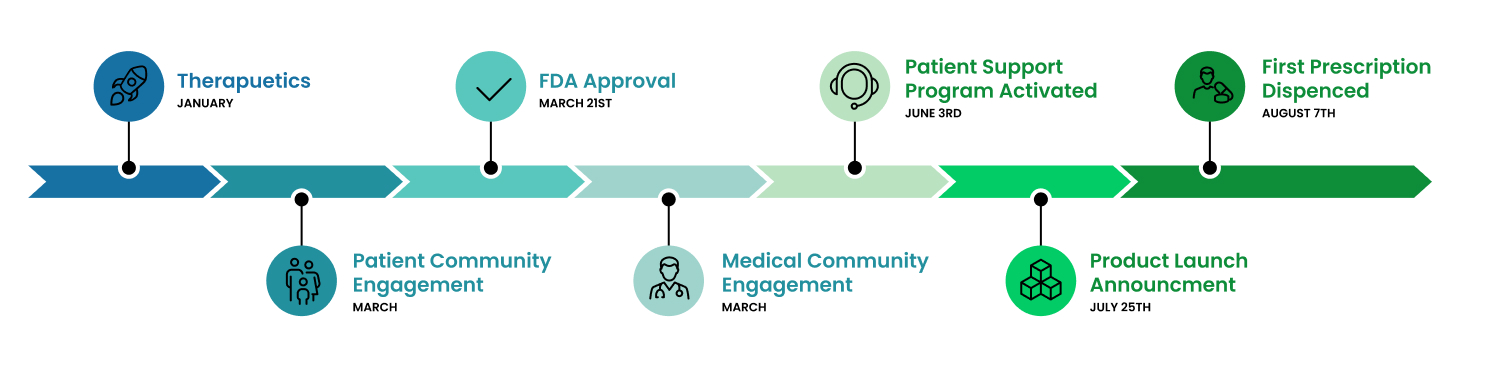
Within seven months of launching ITF Therapeutics, we got a treatment with a novel mechanism of action into the hands of rare disease patients with serious unmet need. Our FDA-approved therapy inhibits histone deacetylase (HDAC), an enzyme that is overactive in certain neuromuscular diseases, rare cancers and other disorders.
This initial product reflects the first chapter in our mission to deliver innovative therapies with urgency, ambition, and compassion for each rare disease community we serve.
Company Launch to Market Access